
FreshPax® Packets
Freshpax Packets come in a grease resistant film and are great for food applications. Can be automated or inserted manually.

FreshPax® CR
FreshPax CR provides your customers with fresh and visually appealing beef, pork, lamb, and value-added meat products.

StabilOx® Canister
StabilOx oxygen absorbing canisters eliminate oxygen to help you achieve better product stability. Up to 5x better performance.

FreshMax®
These expert-designed, adhesive backed oxygen absorbers for food offer a flat, flexible format, with an ultra-thin design.

FreshCard®
Satisfy your customers and reduce shrink by eliminating need for preservatives while maintaining freshness.
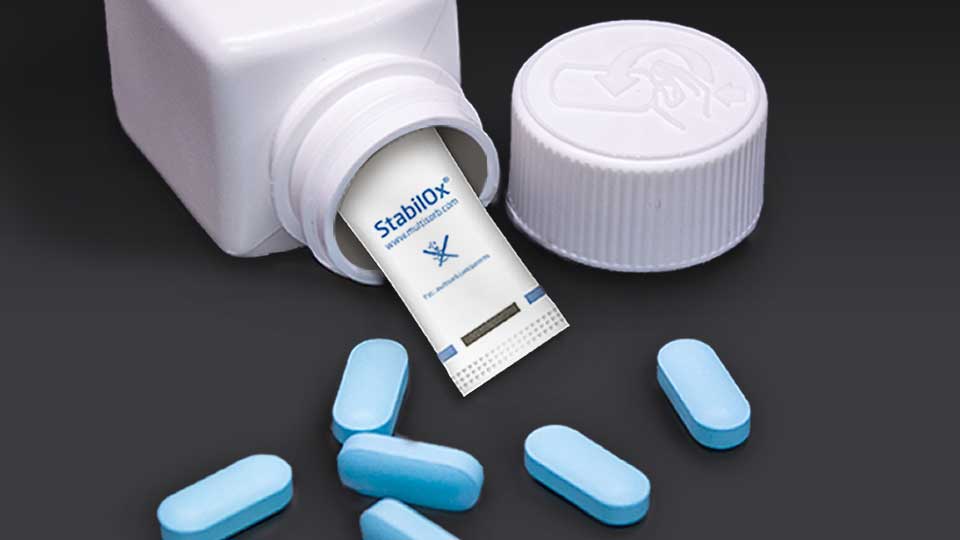
StabilOx® Packet
StabilOx oxygen absorbing packets eliminate oxygen to achieve better product stability. Up to 5x better performance.

